Healthcare App Development Company
Telemedicine, EHR, and mHealth solutions built for startups, clinics, and health systems. We ship production-ready healthcare software across mobile and web.
Top Rated
Clients
Transforming Businesses With Proven Results
Faster development, greater efficiency, trusted globally, and driven by referrals.
savings with AI-powered Engineering
users trust our products
efficiency boost for businesses
of new clients come from referral
HIPAA-Compliant Software Development
HIPAA compliance is an architecture decision, not a feature you add before launch. We define it on day one of every engagement and validate it every sprint.
Telemedicine Application Development
HIPAA-compliant telehealth apps with video/audio consultation (WebRTC), e-prescriptions, appointment scheduling, and secure medical record sharing. Multi-party calls for specialist consultations. Supports iOS, Android, and browser-based access. Integration with EHR systems for seamless clinical workflows.
EHR / EMR Development
Custom Electronic Health Record systems with HL7 FHIR interoperability, clinical documentation, order entry (CPOE), medication management, and clinical decision support. Role-based dashboards for physicians, nurses, and administrative staff. Mobile companion apps for bedside charting.
Remote Patient Monitoring
Platforms that ingest data from wearables (Apple Watch, Fitbit, medical-grade devices), IoT sensors, and patient-reported inputs. Real-time dashboards for care teams, automated threshold alerts, trend analysis, and integration with billing systems for RPM CPT code documentation.
Hospital Information Management System (HIMS)
End-to-end hospital management covering OPD/IPD workflows, bed management, patient registration, billing, pharmacy inventory, lab integration, and MIS reporting. Multi-department dashboards with role-based access.
Health Information Exchange (HIE)
Secure data exchange platforms enabling real-time sharing of patient records, lab results, and referrals between providers, labs, and payers. HL7 FHIR and CDA compliant with consent management and audit logging.
Pharmacy Management Software
Prescription fulfillment workflows, inventory management with reorder alerts, drug interaction checking, controlled substance tracking (DEA Schedule compliance), and integration with insurance/PBM systems for claims processing.
Laboratory Management System (LIS)
Sample accessioning, test ordering, result entry and validation, auto-verification rules, instrument interfacing, and secure result delivery to referring providers. Bar-code driven workflows to reduce manual errors.
Medical Billing Software
Automated charge capture, CPT/ICD-10 coding assistance, claim generation and submission, ERA/EOB processing, denial management, and patient statement generation. Dashboard analytics for revenue cycle KPIs (days in AR, clean claim rate, denial rate).
Medical Device Software
Software development for medical devices following IEC 62304 lifecycle standards. Risk-based classification, software architecture documentation, unit/integration/system testing with traceability, and support for FDA 510(k) or De Novo submissions. Companion apps for connected devices.
Wellness and Fitness Applications
Activity tracking, workout logging, nutrition tracking, and health metric monitoring (heart rate, sleep, steps). Integration with Apple HealthKit and Google Health Connect. Gamification features for user engagement.
Wearable App Development
Apps for smartwatches (Apple Watch, Wear OS), fitness bands, and medical-grade wearables. Real-time data sync, background health monitoring, complications/tiles for at-a-glance data, and low-power BLE communication with companion devices.
Medical Insurance and Claims Applications
Apps connecting patients with insurance providers — plan details, coverage verification, pre-authorization workflows, claim submission and tracking, and explanation of benefits (EOB) access.
Steps to Build a HIPAA-Compliant Healthcare App
01
Define the Use Case and Compliance Scope
Identify the core workflow — telemedicine, RPM, EHR, patient portal, or medical device companion app. Map which regulations apply (HIPAA for US, GDPR for EU, FDA/IEC 62304 for medical devices). Define what constitutes PHI in your system and where it's stored, transmitted, and processed.
02
Design Secure and Intuitive Workflows
Role-based access control (RBAC): patients, providers, admins, and billing staff each see only what they need. HIPAA-compliant UX patterns: session timeouts, auto-lock, biometric authentication, and consent capture before data sharing. Wireframes reviewed against compliance requirements before development begins.
03
Implement Data Security Controls
AES-256 encryption at rest and TLS 1.3 in transit. Secure authentication (OAuth 2.0, MFA, biometrics). Comprehensive audit trails logging every access, modification, and export of PHI. Automated backup with point-in-time recovery. Penetration testing before deployment. Apply encryption, secure authentication, and audit trails to protect patient data.
04
Test, Validate, and Deploy
Compliance validation testing (HIPAA security checklist, FDA design verification/validation). Automated and manual security testing. Deployment to a compliant cloud infrastructure (AWS GovCloud, Azure Healthcare APIs, or Google Cloud Healthcare API). Post-launch monitoring with incident response procedures.
Healthcare Regulatory Compliances We Build For
HIPAA is the baseline. Depending on your product, target market, and device classification, you may also need FDA clearance, IEC 62304 certification, GDPR compliance for EU distribution, or SOC 2 for enterprise contracts.
Health Insurance Portability and Accountability Act (HIPAA)
End-to-end encryption (AES-256), access controls, audit trails, BAA support, breach notification workflows
Food and Drug Administration (FDA)
Risk classification, Design History File, predicate device analysis, 510(k) submission documentation support
Health Information Technology for Economic and clinical Health (HITECH)
Enhanced penalties compliance, breach notification within 60 days, encryption requirements
The General Data Protection Regulation (GDPR)
Data minimization, consent management, right to erasure, DPO support, cross-border transfer safeguards
HL7 / FHIR
FHIR R4 resource mapping, CDA document generation, ADT messaging, lab result exchange
IEC 62304
Software development plans, architecture documentation, unit/integration/system testing per risk class, traceability matrices
The Personal Information Protection and Electronic Documents Act (PIPEDA)
Consent-based data collection, access rights, retention policies
International Medical Device Regulators Forum (IMDRF)
SaMD risk categorization (I-IV), clinical evaluation, post-market surveillance
SOC 2 Type II
Continuous monitoring, access management, incident response, annual audit readiness
Why Healthcare Companies Choose Tech Exactly
Every development agency says they handle HIPAA. Here's what actually separates compliance-first development from compliance-checked-off.
Compliance Built In, Not Bolted On
We don't build the app first and then "make it HIPAA compliant." Compliance requirements (HIPAA, FDA, IEC 62304, SOC 2) are defined during architecture design and validated throughout development. Every sprint includes compliance checkpoints — not just a final audit.
9 Regulatory Frameworks, One Team
Most development agencies handle HIPAA at best. We build for HIPAA, FDA, IEC 62304, HITECH, HL7/FHIR, GDPR, SOC 2, PIPEDA, and IMDRF. Whether you're launching in the US, EU, Canada, or globally — we've mapped the regulatory requirements.
Medical Device Software Experience (IEC 62304)
We build Software as a Medical Device (SaMD) following IEC 62304 lifecycle standards — design history files, risk management per ISO 14971, traceability matrices, and documentation that supports FDA 510(k) submissions. This is specialized work that most app development agencies can't do.
Full-Stack Health Tech Team
Not just mobile developers who read a HIPAA guide. Our health app developers include backend engineers who build compliant cloud infrastructure, security engineers who run penetration testing, and QA engineers who validate against regulatory checklists.
10+ Years, Production Healthcare Apps
We've been building healthcare software since 2014. Our team has shipped telemedicine platforms, EHR systems, RPM solutions, and IEC 62304-compliant medical device software that are in active clinical use.
Healthcare Tech Stack
How We Build Healthcare Apps
Take a Look at Our Case Studies
We’re proud of our work and excited to share how we've helped businesses succeed.
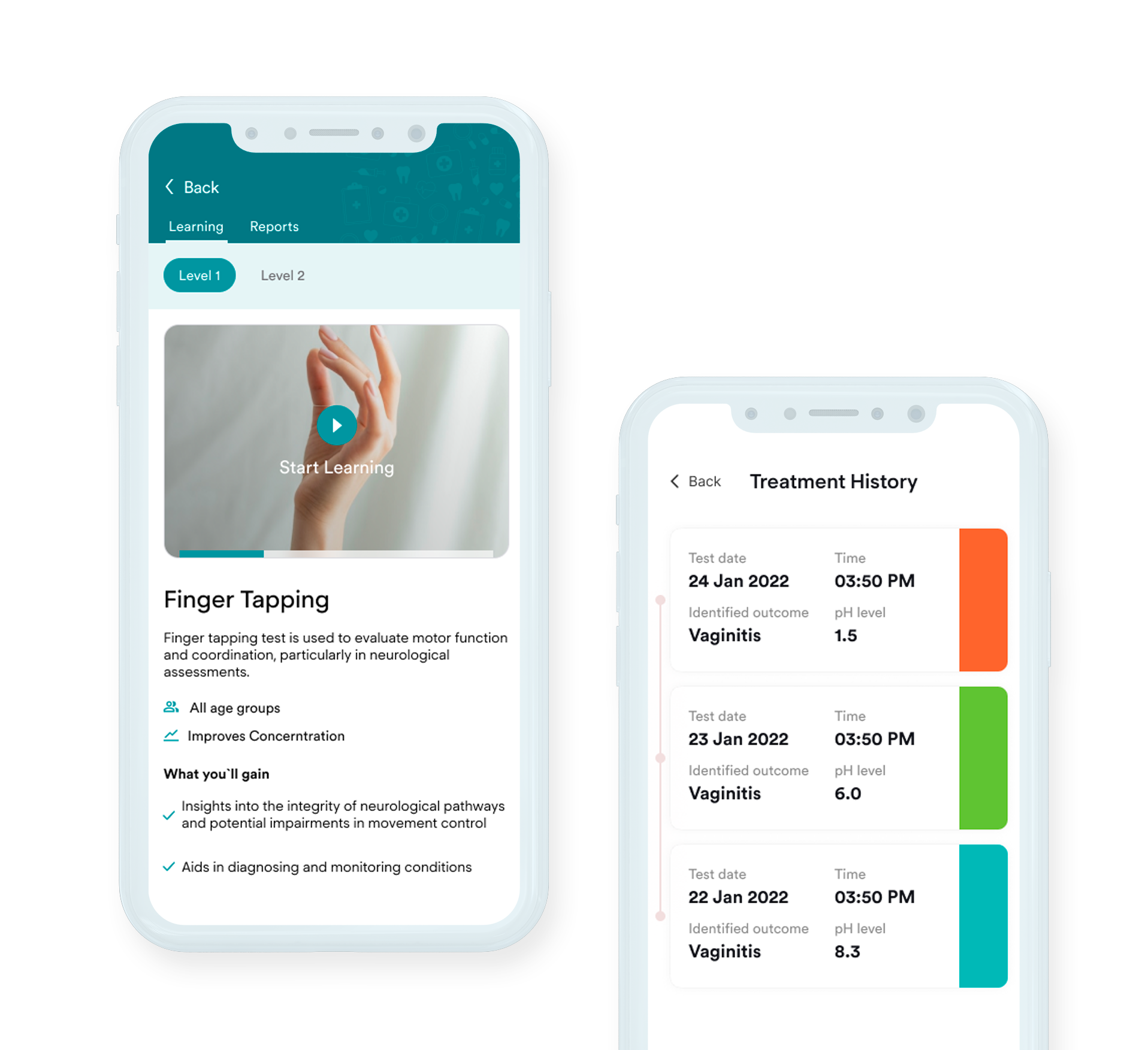
Learn about how we delivered a HIPAA-compliant website that offers online therapy sessions in NYC
Making mental healthcare ready, available, and cheap for everyone by delivering a comprehensive web application in accordance with HIPAA requirements
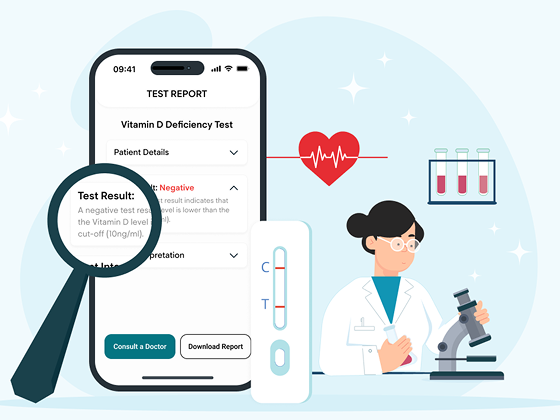
IEC 62304-Compliant Mobile App for Accurate Test Interpretation
A medical-grade mobile app to deliver ~ 70% precise interpretation of home diagnostic tests
Built under IEC 62304 for full compliance
Instant, accurate image analysis
Full traceability and validation
Frequently Asked Questions
A basic patient-facing app (appointment booking, secure messaging, medication reminders) costs $25,000-$50,000. A telemedicine platform with video consultations, e-prescriptions, and EHR integration costs $50,000-$150,000. Complex systems like full EHR/EMR platforms, hospital management systems, or IEC 62304-compliant medical device software can range from $150,000-$500,000+. Our mhealth app development services include detailed discovery, compliance planning, and architecture estimation before development begins.
A patient portal or basic telehealth MVP takes 10-14 weeks. A full telemedicine platform with EHR integration takes 4-6 months. Enterprise hospital management systems or FDA-regulated medical device software can take 8-12+ months including documentation and submission support. Experienced mhealth developers help streamline delivery while ensuring documentation, security, and compliance requirements are properly managed.
Not all. If your app doesn't store, transmit, or process Protected Health Information (PHI), HIPAA may not apply. However, most healthcare apps that interact with patient data — even indirectly — fall under HIPAA. Our mhealth app development services are designed with a compliance-first architecture to reduce long-term security and regulatory risks.
Every HIPAA-compliant build starts with the same baseline: AES-256 encryption at rest, TLS 1.3 in transit, OAuth 2.0 with MFA, role-based access control, comprehensive audit trails on every PHI access/modification/export, session timeouts, and biometric or device-level authentication on mobile. Project-specific controls (HSM-backed key management, dedicated VPCs, automated PHI redaction in logs) are layered on top during architecture design.
There's no such thing as HIPAA certification. HHS doesn't certify software or vendors — any company advertising "HIPAA certified" is either using the term loosely or referencing a third-party attestation (HITRUST, SOC 2 + HIPAA mapping). HIPAA compliance is an ongoing operational state: implementing the required Security Rule and Privacy Rule controls, conducting regular risk assessments, signing BAAs, and being able to demonstrate compliance during an OCR audit if one happens.
If IEC 62304 applies to your product, the documentation requirements alone — traceability matrices, design history files, risk management per ISO 14971 — are significant.
Yes. We integrate with major EHR platforms (Epic, Cerner, Allscripts, athenahealth) using HL7 FHIR R4 APIs, and with custom/legacy systems using HL7 v2 messaging, CDA documents, or direct database integration. FHIR is the modern standard — if your EHR supports FHIR, integration is significantly faster and more maintainable.
Our mhealth app development services cover iOS, Android, and cross-platform development with HIPAA compliance, HealthKit/Health Connect integration, and enterprise-grade security built in.
Multiple layers: AES-256 encryption at rest, TLS 1.3 in transit, OAuth 2.0 with MFA for authentication, role-based access control (RBAC), comprehensive audit trails, session timeout policies, device-level security (biometrics, secure enclave/keystore), and regular penetration testing. We also implement automated threat monitoring and incident response procedures.
We support the software development and documentation side of FDA submissions. This includes Design History Files, risk management documentation (ISO 14971), software verification and validation testing, traceability matrices, and cybersecurity documentation. We work alongside your regulatory affairs team or regulatory consultants to prepare the technical package for 510(k) or De Novo submissions.
Ready to Get Started?
Get a free quote and see what we can do for you.
