Healthcare App Development in 2026: What Actually Goes Into Building a Compliant Product

- Compliance isn’t a layer you add later; your entire architecture must be HIPAA-compliant from day one, since a single overlooked provision can result in fines up to $2.1 million per violation category annually.
- Regulatory missteps go beyond HIPAA, a missed IEC 62304 classification can stall an FDA submission for months, and EHR integrations involving legacy HL7v2 systems can consume half your budget if not planned for upfront.
- There’s no such thing as “HIPAA certification”; what exists is HIPAA compliance, validated through third-party audits like HITRUST or SOC 2 Type II. Anyone selling you a certification badge is not delivering an actual standard.
- Understanding the clinical environment is just as critical as writing good code; teams that ignore how healthcare workflows actually function will see that gap reflected directly in the final build.
Healthcare app development seems quite easy until you really do it. On paper, it all makes sense: you nail down your features, put a team together, and get an MVP out the door. We have worked on enough projects to be bluntly honest that this is far from reality.
The reality is that healthcare app development is one of the most regulation-dense, technically demanding areas of software development. A single overlooked HIPAA provision can lead to fines up to $2.1 million per violation category annually. A missed IEC 62304 classification can stall your FDA submission for months. An EHR integration that looked simple in the architecture diagram can eat through half your budget if you haven’t accounted for HL7v2 legacy systems that most hospitals still run.
There is a whole other side to this challenge, too. The complex systems and strict regulations create a strong barrier. Once you can meet compliance requirements, integrate with EHR legacy systems, and build something that doctors and clinicians genuinely use — ideally with the help of an expert healthcare app development company like Tech Exactly — you will be left with a defensible product. In many ways, the struggle is what makes the potential payoff so significant.
For over ten years, Tech Exactly has been building healthcare apps. We’ve built everything from telemedicine platforms and EHR systems to remote patient monitoring tools. Drawing from that experience, we have created this guide that outlines the true lifecycle of healthcare app development. We will walk you through everything from the critical compliance decisions you need to make before writing any code, to the post-launch monitoring that keeps your app out of regulatory nightmares.
Our healthcare app development guide covers the different types of healthcare apps and the features they typically include, if that’s what you are looking for. This article is a breakdown of the brutal reality of the process: the sequence of decisions, technical trade-offs, and compliance strategies that decide whether your healthcare app actually ships or dies in development.
If you are a developer who is looking to understand why it is harder to build a healthcare app than regular software, this guide is for you.
Why Healthcare App Development Doesn’t Follow Standard Software Rules

The basic development flow looks largely the same as building any other software project. This part is no mystery: you define requirements, design the product, build it, test it, and deploy it.
What really makes healthcare different is that there are three additional layers on top of that process. These layers can be surprisingly complex, and many teams do not fully anticipate them until they are already deeply entangled in them.
The Compliance Layer
The quickest way to get your compliance architecture wrong is to treat it as something you could figure out later.
Any healthcare application that handles Protected Health Information (PHI) needs HIPAA compliance. The main part is that HIPAA compliance is not just a one-time task; it is a rigid set of administrative, physical, and technical safeguards that need to be hardwired into your architecture from the first day. It is essential to include AES-256 encryption for data at rest and in transit, precise role-based access controls, multi-factor authentication, automatic session timeouts, and exhaustive audit logging for all data access.
If your application interprets medical data, makes clinical decisions, or directly impacts patients’ treatments, it will qualify as software as a medical device (SaMD). This would mean that you are now dealing with FDA oversight and IEC 62304 compliance. That’s a completely different regulatory regime with its own documentation, risk classification, and verification requirements.
The Integration Layer
A key point to understand is that a healthcare app can connect to multiple systems to be really useful; they don’t operate in a vacuum. This could include. EHRs, pharmacy platforms, lab systems, insurance processors, wearables.
The real challenge is that none of these systems speak the same language. Each uses different standards: FHIR, HL7v2, X12 for claims, and NCPDP for pharmacy data.
There is a huge difference between “we support FHIR” and “we integrate seamlessly with Epic, Cerner, and Meditech in production”. Honestly, most of the EHR integration projects we take on are rescue missions. We often step in after another team has failed to understand and underestimated the chaos
The Clinical Workflow Layer
You have to remember the end user of the app. They are not sitting idle at a desk. They’re nurses during shift handoffs, physicians between patient consultations, and lab technicians processing dozens of samples. If your app adds friction to an existing workflow, even an extra thirty seconds, it won’t get adopted.
This is the struggle point for most general software firms. Just writing good code is not enough in healthcare app development. One must understand how the clinical environment works, as they have their own logic, their own pressures, and it is just as important as the code. If the development team does not focus on this, it will reflect in the final build.
Let's Start Your Project Today
Ready to build your healthcare app with us? Reach out now – our experts are just one click away.
The Healthcare App Development Process: How It Actually Works
Below is how the real process works. Not the delusional version but the one that accounts for compliance reviews, integration headaches, and the regulatory realities of building a software that handles people’s medical data.
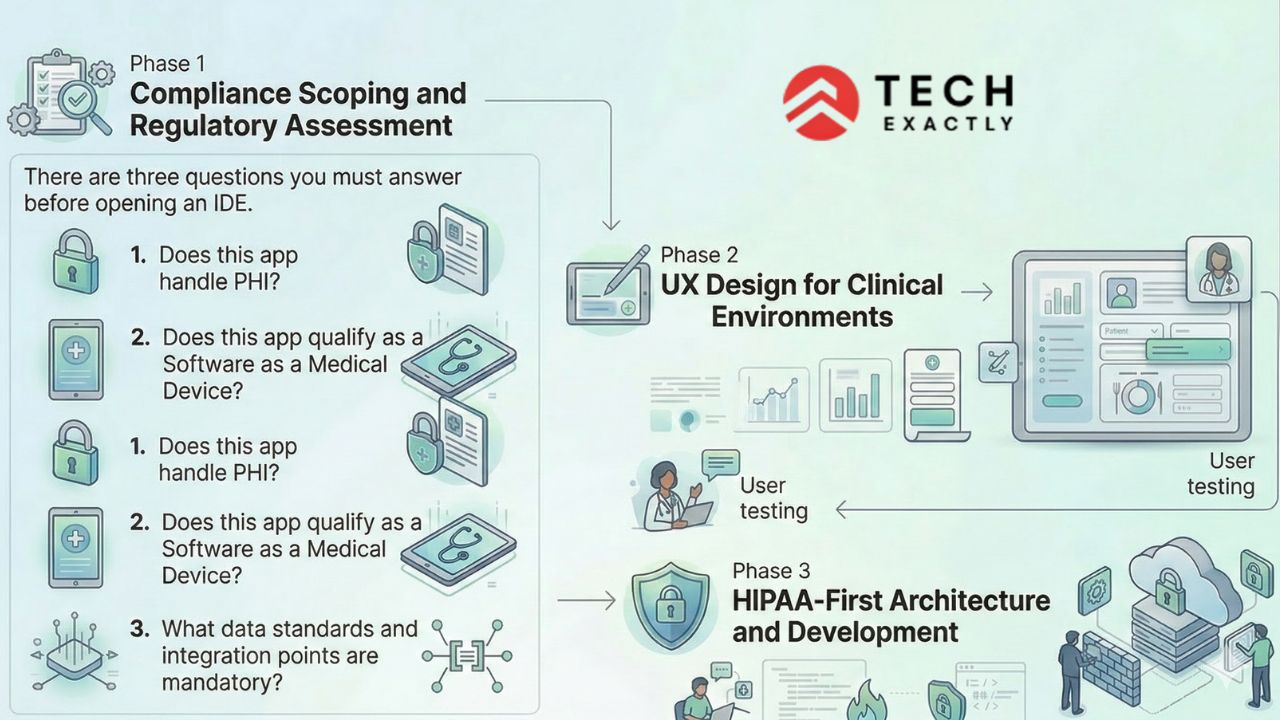
Phase 1: Compliance Scoping and Regulatory Assessment
There are three questions you must answer before opening an IDE.
1. Does this app handle PHI?
If yes, and the answer is almost always yes, your entire architecture must be HIPAA-compliant. That doesn’t mean an extra security layer that you need to be compatible with later. It means your database design, API structure, hosting environment, and access controls are all built around HIPAA from the beginning.
And what if it doesn’t handle PHI? In 2026 ‘We aren’t a HIPAA-covered entity’ this doesn’t work anymore. If you are creating an app for the consumer’s health or wellness, you fall under the FTC’s Health Breach Notification Rule (HBNR).
Sharing consumer health data with marketing pixels like Meta or Google without clear consent will trigger massive FTC fines. Additionally, state laws like Washington’s My Health My Data Act (MHMDA) have created a surge of private class-action lawsuits for apps that mishandle consumer health metrics. The bottom line is simple: if your product touches health data in any way, there is no option other than having a compliance strategy, period.
2. Does this app qualify as a Software as a Medical Device?
The FDA follows a strict risk-based classification system. If the application is limited only to administrative work such as scheduling, billing, or communication, then it would be exempted. But if it assists with clinical decision making, even if it interprets medical data or controls any medical device, then it is likely to fall into the Software as a Medical Device (SaMD) category.
The classification matters because it determines whether you need a 510(k) submission, De Novo authorization, or Pre-Market Approval (PMA), each with very different timelines and costs.
3. What data standards and integration points are mandatory?
Every external system that your app needs to communicate with must be mapped out. EHR systems? Which ones? Which FHIR version do they support? Are HL7v2 interfaces still required for specific data types? What about state-level immunization registries or prescription drug monitoring programs?
This phase usually takes around 3-5 weeks for a reasonably complex healthcare application. If you skip or rush through it, it can become one of the costliest mistakes in healthcare app development. There are a lot of projects that end up exceeding their budgets by 40–60% only because major architectural changes happen due to compliance requirements being discovered late in the process.
Phase 2: UX Design for Clinical Environments
Healthcare UX is not a consumer app UX. The constraints are fundamentally different:
Context matters. There are different attention constraints for a surgeon reviewing imaging results pre-operation than a patient checking lab results from home. They both might use the same app, but they will need radically different interfaces
Error tolerance is near zero. In an e-commerce app, a misclick means that someone adds the wrong item to a cart. In a healthcare app, a mistap could end up as a wrong medication dose or a missed critical alert. Interface design has to account for high-stakes, low-attention environments.
Accessibility isn’t optional. Healthcare apps typically serve elderly patients, people with visual impairments, individuals with cognitive disabilities, and clinicians wearing gloves or using screens in bright operating rooms. WCAG 2.1 AA compliance is the baseline, not a nice-to-have.
What works in practice: Role-based dashboards (a nurse sees different information than a physician), progressive disclosure (show critical info first, details on demand), oversized touch targets for mobile use in clinical settings, and high-contrast modes for varying lighting conditions.
The design phase should run alongside compliance scoping, and it takes around 4-6 weeks. Workflows are mapped with real clinical users and not personas or assumptions, but through real and direct observation of how healthcare professionals actually interact with already existing systems during this stage.
Phase 3: HIPAA-First Architecture and Development
Here’s where custom healthcare app development diverges most sharply from standard development.
Infrastructure decisions come first. Before anything else, you must decide on an architecture. A HIPAA-eligible infrastructure is a must-have, which in practice means AWS with a signed Business Associate Agreement, Azure Healthcare APIs, or Google Cloud Healthcare API.
A lot of people miss out on the fact that not all services on these platforms are HIPAA-eligible. For example, on AWS, RDS and S3 with encryption are HIPAA-eligible, but not every AI or ML service falls under their BAA. This is an important distinction, and it needs to be checked service by service before your architecture is finalized.
The security architecture is non-negotiable:
- Encryption: AES-256 for data at rest. TLS 1.2+ for data in transit. No exceptions.
- Authentication: Multi-factor authentication (MFA) for all clinical users. OAuth 2.0 or SAML for single sign-on with hospital identity providers.
- Access controls: Role-Based Access Control (RBAC) at minimum. Many organizations now require Attribute-Based Access Control (ABAC) for finer-grained permissions — a physician in cardiology should see cardiac patient records, not psychiatric records.
- Audit logging: Every access to PHI must be logged — who accessed what, when, from where, and what they did with it. These logs themselves need to be tamper-proof and retained for a minimum of six years under HIPAA.
- Session management: Automatic timeout after inactivity. Session tokens that can’t be reused across devices.
Development methodology. We use agile sprints with a compliance checkpoint at every sprint review. Each user story has acceptance criteria that include security requirements, not just functional requirements. “As a physician, I can view patient lab results” isn’t complete. It needs “…after authenticating with MFA, with access logged, and results encrypted during transmission.”
This phase is the longest, typically 12-20 weeks, depending on complexity. During this, the development and compliance teams must be in constant communication. We’ve found that embedding a compliance specialist directly in the development team, rather than having them review work after the fact, reduces rework.
If you are a healthcare startup, follow our guide on how to build a regulatory-compliant IEC 62304 mobile app without slowing you down.
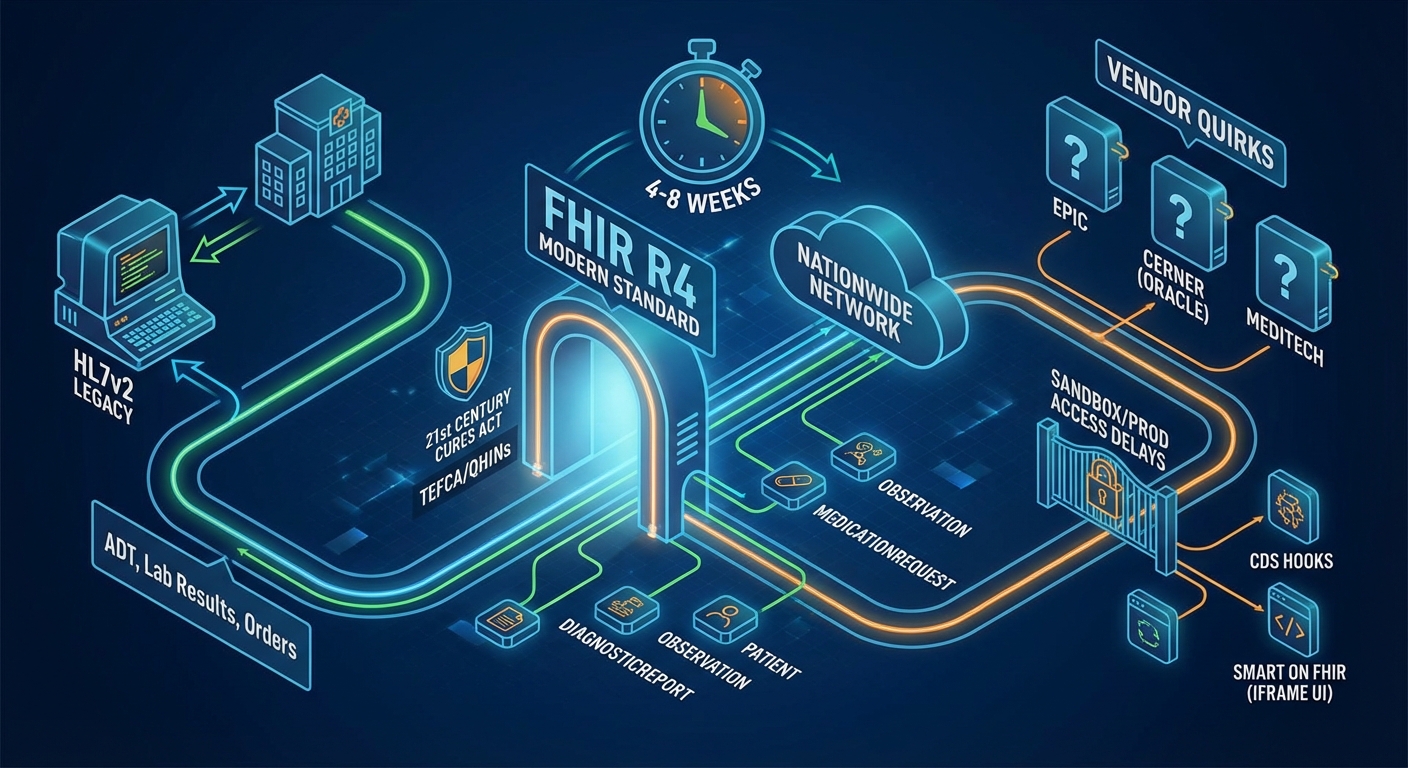
Phase 4: EHR and Third-Party Integration
This is the part where the timeframe starts feeling unpredictable.
FHIR (Fast Healthcare Interoperability Resources) is the modern standard, and the 21st Century Cures Act is pushing all US healthcare systems toward it. Beyond basic FHIR, 2026 interoperability is defined by TEFCA (the Trusted Exchange Framework and Common Agreement). Your architecture needs to anticipate connecting to Qualified Health Information Networks (QHINs).
We are moving away from point-to-point EHR integrations and toward a nationwide network model. If your integration strategy is just ‘build an API and hope Epic approves it,’ you are designing for the past.
The good news: if your integration partner supports FHIR R4, data exchange is relatively standardized for common resources like Patient, Observation, MedicationRequest, and DiagnosticReport.
However, many hospital systems still rely on HL7v2 messaging for ADT (Admit–Discharge–Transfer) events, lab results, and clinical orders. In practice, you’ll often need to support both: a modern FHIR API layer for newer systems and an HL7v2 integration engine for legacy hospital infrastructure, which are not going away anytime soon.
Practical integration challenges we run into:
- Epic, Cerner (now Oracle Health), and Meditech each have their own FHIR implementation quirks. What works in one doesn’t always work in another without adjustment.
- Getting sandbox access for development takes time, sometimes weeks. Getting production credentials takes even longer.
- CDS Hooks (Clinical Decision Support Hooks) for real-time decision support during clinical workflows add another layer of integration complexity.
- SMART on FHIR apps need to work within the EHR’s existing interface, which means adapting your UI to render inside an iframe or embedded view.
Budget 4-8 weeks for EHR integration, depending on the number of systems and whether you’re dealing with FHIR-native systems or legacy HL7 environments.
Phase 5: AI and Machine Learning Integration
Generative AI and machine learning are becoming table stakes in mobile app development. But integrating AI into a healthcare application is fundamentally different from adding a chatbot to a consumer app.
Clinical AI needs explainability. A model that flags a potential diagnosis needs to show why it reached that conclusion. Black-box predictions aren’t acceptable in clinical settings. This is for regulatory reasons and also because clinicians will neither trust nor use recommendations they can’t understand.
Since 2026, the law has made this explainability mandatory. Under the ONC’s HTI-1 and HTI-2 rules, any clinical application that uses predictive AI or Decision Support Interventions (DSI) and connects to certified EHRs must provide an ‘AI nutrition label.’ There needs to be clear transparency for the users to be able to know exactly what data the model was trained on, its known biases, and its validation metrics. Black-box AI is considered dead in US clinical settings.
Data governance is critical. It requires careful de-identification, consent management, and often IRB (Institutional Review Board) approval to train or fine-tune models on patient data. You cannot just feed an LLM clinical and hope to get by.
Where AI adds real value in healthcare apps:
- Clinical documentation: Ambient listening that converts physician-patient conversations into structured clinical notes, reducing the burden of documentation
- Diagnostic support: Image analysis for radiology, pathology, and dermatology, always as decision support, not decision replacement
- Predictive analytics: Risk stratification for readmission, sepsis early warning, and medication adherence prediction
- Patient engagement: Intelligent symptom triage, medication reminders with contextual health education, and personalized care plan recommendations
You are in the SaMD territory if your app includes AI features that influence clinical decisions. Plan your regulatory strategy according to that.
Phase 6: Testing Beyond Functional QA
If your QA process looks similar to other basic software, then it is not thorough enough; you are lagging. Unit tests, integration tests, UAT, all of it still needs to happen. But when it comes to healthcare, those are the foundation and not the ceiling.
Security testing includes penetration testing by a third-party firm (required for most healthcare organizations), vulnerability scanning, and social engineering testing. We typically engage a HITRUST-certified security assessor.
Compliance validation involves a formal review against HIPAA’s technical safeguards, documented in a compliance matrix that maps each requirement to the specific technical control that addresses it.
If you are building Software as a Medical Device (SaMD), compliance is no longer just an option. Under Section 524B of the FD&C Act, the FDA will reject your submission if you cannot provide a complete Software Bill of Materials (SBOM).
Additionally, you also need to have a clear, documented plan giving an explanation about how you’ll monitor, detect, and fix cybersecurity vulnerabilities after the product is on the market. Its not enough to show that your software is secure today; you also need to prove how you will keep it secure in the upcoming years.
Clinical validation means putting your app in front of real clinicians in a controlled environment and watching them use it. It will not be a demo, but a real simulation with realistic patient data. This catches workflow problems that no amount of automated testing will find.
Interoperability testing confirms that your EHR integrations work not just in sandboxes, but with production-like data volumes and edge cases. HL7v2 messages from different EHR vendors can have subtle formatting differences that only surface with real-world data.
In our experience, this phase usually adds at least 4-6 weeks to the timeline. There are a lot of teams that try to squeeze it or run it in parallel with development in order to save time. Unfortunately, this often results in compliance gaps that only surface later, sometimes after launch, when regulatory reviews reveal issues that should have been addressed earlier.
Phase 7: Deployment and Post-Launch
The Deployment environment must match your HIPAA-compliant architecture. Container orchestration with Kubernetes on a HIPAA-eligible cloud, with infrastructure-as-code for reproducible, auditable deployments.
HIPAA compliance doesn’t only apply to tour production environments. Every environment, such as Staging and disaster recovery which touches the patient’s data has to meet the same compliance standards. There are no exceptions.
Post-launch isn’t maintenance, it’s continuous compliance. HIPAA requires ongoing risk assessments (at least annually, but we recommend quarterly). Software dependencies need security patching. Access logs need regular review. If regulations change, and they change often, then your app needs to adapt.
Monitoring specifics:
- There needs to be real-time alerts for unauthorised attempts to access the data
- Performance monitoring to catch degradation that could affect clinical workflows
- Uptime monitoring with escalation protocols, because it is very different for an e-commerce website to go down than for a healthcare app to go down during a hospital’s peak hours.
- Regular backup testing: not just running backups, but actually restoring from them to verify integrity
Let's Start Your Project Today
Ready to build your healthcare app with us? Reach out now – our experts are just one click away.
Key Technology Decisions in Healthcare App Development
Every healthcare app development project will involve a few critical tech choices. Here is what we think about it
Native vs. Cross-Platform
Native (Swift/Kotlin) will give you the best performance and also provide access to device-specific features like HealthKit (iOS) and Health Connect (Android). Native is often the right call for apps that interact with medical devices via Bluetooth LE or need real-time biometric data streaming
Cross-platform (React Native or Flutter) makes sense when you need to ship on both iOS and Android quickly, and the app is primarily for displaying dat, communication, or workflow management. Most telemedicine apps and patient portal applications work well cross-platform.
We use React Native for most of our healthcare projects. The shared codebase alone brings the development time down, and in our experience, the performance difference with native apps hardly matters for the kind of use case and functionality most healthcare apps need
Cloud Provider
All three major providers — AWS, Azure, and Google Cloud — offer HIPAA-eligible services with BAA coverage. The choice usually comes down to existing infrastructure:
- AWS has the broadest set of HIPAA-eligible services and the largest ecosystem. Most healthcare organizations we work with are already on AWS.
- Azure has strong healthcare-specific offerings (Azure Health Data Services, FHIR Server) and integrates well with organizations already in the Microsoft ecosystem.
- Google Cloud has the Healthcare API and strong AI/ML capabilities, but a smaller footprint in enterprise healthcare.
AI/ML Stack
If you’re embedding AI features, the main decision is build vs. integrate. If you fine-tune an open-source medical LLM, it will give you more control, but it will also require significant ML engineering. If you use managed services such as Azure AI Health Insights or AWS HealthLake, then it will get you to market much faster, but there will be less customisation.
For most projects, we recommend a Retrieval-Augmented Generation (RAG) approach: keeping your LLM general-purpose but grounding its responses in your organization’s clinical knowledge base. It’s more maintainable, more auditable, and doesn’t require the massive datasets needed for full fine-tuning.
The Mistakes That Actually Sink Healthcare App Projects
In our experience of more than a decade of building healthcare applications, these are the patterns we see in most projects that fail.
Treating compliance as a later-stage concern. If your database schema does not support field-level encryption and audit logging from the first day itself, retrofitting it is a partial rewrite. There are so many times when we were hired just because the original team could not build a functional app that passes a HIPAA audit.
Underestimating EHR integration. We budget 20-30% of the total project effort for integration work. There are teams that estimate only around 10% and end up scrambling.
Ignoring the 21st Century Cures Act. Since April 2021, information blocking rules require healthcare apps to support patient access to their data. If your app restricts data portability, you’re not just losing users, you’re potentially violating federal law.
Building for the demo, not the workflow. A demo that amazes the boardroom and a workflow that works on the hospital floor are two different things, and teams often confuse them more than you would think. If even a single feature adds friction for a nurse or the physician, then it hardly matters how well it is tested in the conference room, as it will not be used.
Skipping the Business Associate Agreement. Every third-party service that touches PHI — be it your cloud provider, your analytics tool, your crash reporting service — they need a signed BAA. We have seen organizations use consumer-grade tools such as standard Google Analytics and Mixpanel without even realizing it the least bit that they were in violation.
What Comes Next in Your Healthcare App Development Journey
We are not going to feed you the lie that healthcare app development is easy or is getting easier, and anybody saying otherwise is just not paying attention. The FDA is till fine tuning the guidelines to handle AI-enabled medical devices. The Cures Act is pushing interoperability requirements further than most teams are even ready for. And also, the patients who use consumer apps every day are bringing those same expectations into digital health. The bar is moving, and it is moving in one direction.
The organizations that succeed are the ones that treat compliance and clinical usability as core product requirements, not afterthoughts. They invest in the upfront scoping that prevents expensive pivots later. And they work with development teams that have actually shipped healthcare software into production — not teams learning healthcare regulations on your project’s timeline.
If you are planning a healthcare app and want to be sure that it will survive the 2026 regulatory landscape, don’t guess. We offer a 2026 Compliance & Architecture Blueprint session. We will map out the specific FTC/HIPAA liabilities, EHR integration bottlenecks, within a timeline that is realistic to get the product into clinical hands
Let's Start Your Project Today
Ready to build your healthcare app with us? Reach out now – our experts are just one click away.
FAQs About Healthcare App Development
For a not-too-complex healthcare application, like a telemedicine platform with EHR integration and HIPAA compliance, expect 6-9 months from initial scoping to production launch. Simpler apps, such as patient portals and appointment scheduling, can be finished in 3-5 months. Medical device software with FDA submission requirements normally take about 9-14 months.
Ranges widely based on complexity. A basic HIPAA-compliant patient engagement app starts around $80K-$120K. A full telemedicine platform with EHR integration runs $150K-$300K. Medical device software with regulatory submission support can exceed $400K. We break this down in detail in our healthcare app development cost guide.
There is no such thing as HIPAA certification. What really exists is HIPAA compliance, which means that one must meet the requirements of the Privacy Rule, Security Rule, and Breach Notification Rule. Third-party audits like HITRUST or SOC 2 Type II give you external validation, but there is no official certification that the app is HIPAA compliant. If someone is selling you a certification, all they are doing is selling a badge, not a standard
No. The FDA regulates Software as a Medical Device (SaMD): apps that perform clinical functions like diagnosis, treatment recommendations, or medical device control. Administrative apps (scheduling, billing, patient communication) and general wellness apps are typically exempt. The FDA’s Clinical Decision Support guidance document outlines the specific criteria.
It depends on what the AI does. If AI is used for operational efficiency (scheduling optimization, billing coding assistance), likely no FDA oversight is needed. If it’s used for clinical decision support that clinicians are expected to act on, or diagnostic image analysis, FDA oversight applies. The FDA’s AI/ML-based SaMD Action Plan outlines the evolving framework.
Epic and Oracle Health cover approximately 60% of the US hospital market amongst themselves. Now, if you add Meditech, Athenahealth, and eClinicalWorks, it will fill out most of the rest area left to cover. You must not make the mistake of trying to integrate with all of them in your first version. Instead, figure out the systems your actual target users are running and start there.
Let's Start Your Project Today
Ready to build your healthcare app with us? Reach out now – our experts are just one click away.
Manas Das, Mobile App Architect at Tech Exactly, has over 9 years of experience leading teams in iOS, Android, and cross-platform development. He specialises in scalable app architecture and GenAI-driven mobile innovation.
